 
However, the model assumed electrons in the shells didn't interact with each other and couldn't explain why electrons seemed to stack in an irregular manner. It also explained why the noble gases were inert and why atoms on the left side of the periodic table attract electrons, while those on the right side lose them. 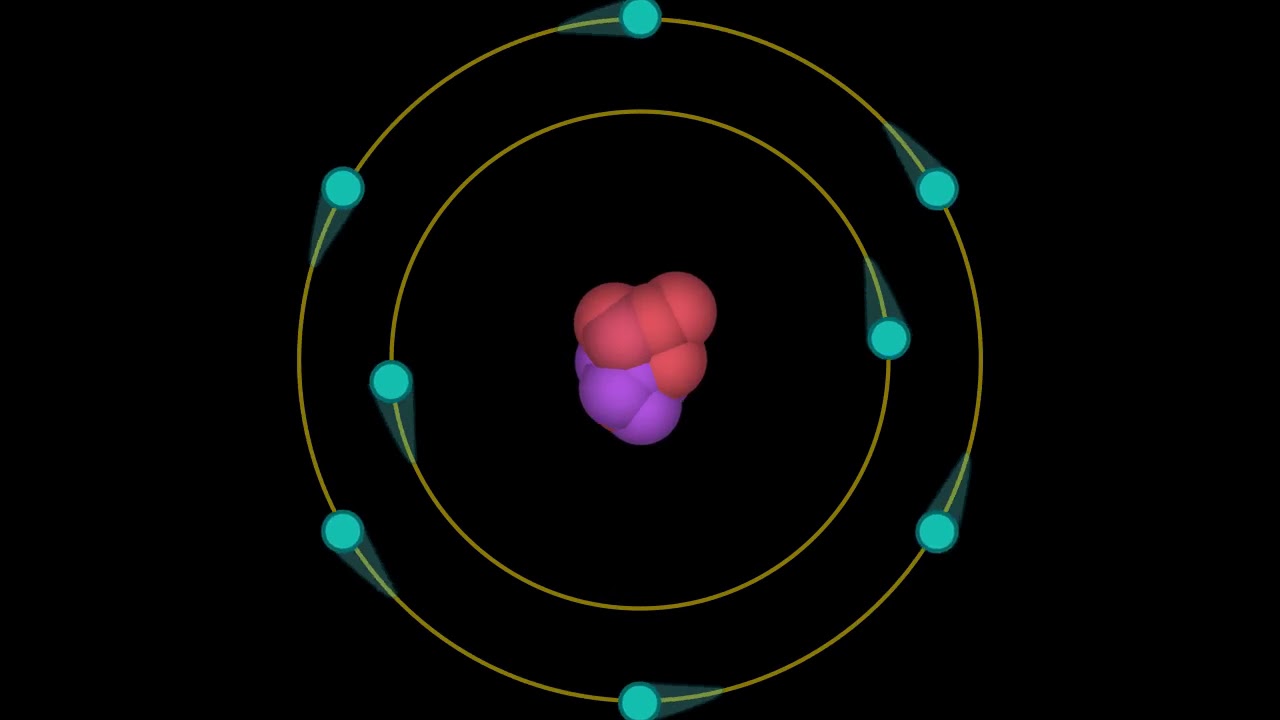
For example, the shell model explained why atoms got smaller moving across a period (row) of the periodic table, even though they had more protons and electrons. Zu jeder dieser Bahnen gehört eine bestimmte konstante Gesamtenergie, die sich als Summe der kinetischen und der potentiellen Energie des. Bahnen, auf denen das Elektron Energie verliert. 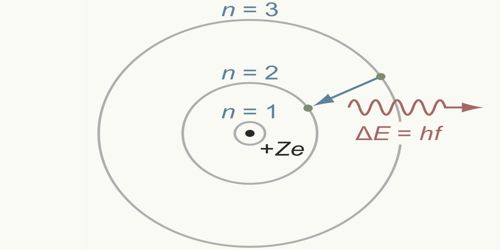
ein 'Axiom', das dazu führt, daß es einige wenige stabile Bahnen des Elektrons um den Atomkern gibt, d.h. The model explained some of the atomic properties of heavier atoms, which had never been reproduced before. Wir wollen nun mit dem Bohrschen Atommodell, welches wir bereits im Abschnitt zur quantenhaften Emission und Absorption kennengelernt haben, einige Berechnungen durchführen. 2.1.2 Fruehe Atommodelle: Bohr und de Broglie. For example, an electrically neutral helium atom has an atomic number Z 2. Hydrogen-like ions are atoms of elements with an atomic number Z larger than one ( Z 1 for hydrogen) but with all electrons removed except one. Thus, the Bohr model for heavier atoms described electron shells. Bohr’s model of the hydrogen atom also correctly predicts the spectra of some hydrogen-like ions. Once the level was full, additional electrons would be bumped up to the next level. Bohr believed each electron orbit could only hold a set number of electrons. More electrons were required to cancel out the positive charge of all of these protons. Heavier atoms contain more protons in the nucleus than the hydrogen atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
